Humans are composed of molecules supplied by Nature's systems engineers ..... that engage in physical biology engineering, brain engineering, human behavior engineering, thought engineering, English language symbols engineering, science equations and formula, and social engineering.
These various projects of Nature are applied to experimental subsets of human. Actual testing is common science lab protocol ..... Nature uses EARTH LAB with humans and human groups.
Let's look at a chemistry molecule expression FORMAT ---> social chemistry and molecule social engineering ERROR signals.
The source of anger ....... covalent bonds in Illinois and Northern Illinois
Chemical Bonds
Covalent bonds, Ionic bonds, Hydrogen bonds - Chemistry | WyzAnt ...
Covalent bond
A covalent bond is the chemical bond that involves the sharing of pairs of electrons between atoms. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding.[1] For many molecules, the sharing of electrons allows each atom to attain the equivalent of a full outer shell, corresponding to a stable electronic configuration.
Covalent bonding includes many kinds of interaction, including σ-bonding, π-bonding, metal-to-metal bonding, agostic interactions, and three-center two-electron bonds
In the molecule H2, the hydrogen atoms share the two electrons via covalent bonding.[5] Covalency is greatest between atoms of similar electronegativities. Thus, covalent bonding does not necessarily require the two atoms be of the same elements, only that they be of comparable electronegativity. Although covalent bonding entails sharing of electrons, it is not necessarily delocalized.
Earth LAB site --> CH = Cole Hall
Department of Chemistry & Biochemistry - NIU - Northern Illinois ...
Course Offerings
CHEM Course Descriptions
Transfer Courses
- Molecule .... Articulation symbol Tables
- Dual Admissions --> additional missions of chemistry department molecules
- Transfer Center to other formats in other dimensions of REALITY of the 11-dimensions of string theory
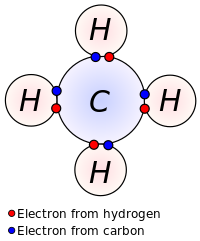
The term "covalence" in regard to bonding was first used in 1919 by Irving Langmuir in a Journal of the American Chemical Society article entitled "The Arrangement of Electrons in Atoms and Molecules". Langmuir wrote that "we shall denote by the term covalence the number of pairs of electrons that a given atom shares with its neighbors."[6]
The idea of covalent bonding can be traced several years before 1919 to Gilbert N. Lewis, who in 1916 described the sharing of electron pairs between atoms.[7] He introduced the Lewis notation or electron dot notation or Lewis dot structure in which valence electrons (those in the outer shell) are represented as dots around the atomic symbols. Pairs of electrons located between atoms represent covalent bonds. Multiple pairs represent multiple bonds, such as double bonds and triple bonds. An alternative form of representation, not shown here, has bond-forming electron pairs represented as solid lines.
Lewis proposed that an atom forms enough covalent bonds to form a full (or closed) outer electron shell. In the methane diagram shown here, the carbon atom has a valence of four and is therefore surrounded by eight electrons (the octet rule); four from the carbon itself and four from the hydrogens bonded to it. Each hydrogen has a valence of one and is surrounded by two electrons (a duet rule) - its own one electron plus one from the carbon. The numbers of electrons correspond to full shells in the quantum theory of the atom; the outer shell of a carbon atom is the n=2 shell which can hold eight electrons, while the outer (and only) shell of a hydrogen atom is the n=1 shell which can hold only two.
While the idea of shared electron pairs provides an effective qualitative picture of covalent bonding, quantum mechanics is needed to understand the nature of these bonds and predict the structures and properties of simple molecules. Walter Heitler and Fritz London are credited with the first successful quantum mechanical explanation of a chemical bond, specifically that of molecular hydrogen, in 1927.[8] Their work was based on the valence bond model, which assumes that a chemical bond is formed when there is good overlap between the atomic orbitals of participating atoms. These atomic orbitals are known to have specific angular relationships between each
Covalent bond
Co....... valent ...... bond
Cole valentine's day
.... molecule mass massacre
Northern Illinois University shooting - Wikipedia, the free encyclopedia
Perceive the building as a left HALL classroom and a right HALL classroom --->
the parallel equivalent at a atom/molecule LEVEL .......
left H = Hydrogen and a right H = Hydrogen.
What's this?
- When two hydrogen atoms get close enough, the electron from each atom feels an attraction from the proton in the other atom's nucleus.
- This attraction pulls the atoms together.
- The electrons end up being shared by the atoms in a region around the nucleus of both atoms.
Covalent Bond in Hydrogen II

- Hydrogen atoms are close together. The electron from each atom feels the attraction from the proton in the nucleus of the other atom.
- This attraction pulls the atoms together and the electrons are shared by both atoms.
- The atoms bond because there is a strong enough attraction in both directions and room for the electrons in the outer energy level of the atoms.
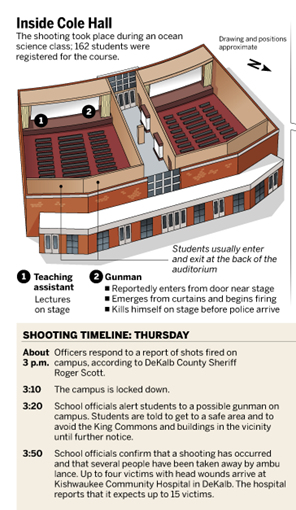
This univeristy buildng in DeKalb, Illinois can be theoretically seen by the UNIVERSE view of EARTH .... as EARTH LAB and a chemistry lab ... in physical chemistry and social chemistry.
Nature's SYMBOL PROCESSOR can equate these 2 lecture Halls to H = Hydrogen (that is ... Cole HALL is the larger scale model, the amplified version of hyfrogen atom). Thus the perception process provided by NATURE ...... Hydrogen atoms VIA the more visible, obvious .... eye/optical image of the cement/ conrete Hall model ..Cole Hall.
Symbolism - Merriam-Webster Online
by expressing the invisible or intangible by means of visible
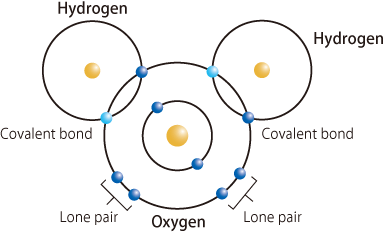
Covalent Bonding in Water I ---> oceanography class was in session.
What's this?
- When two hydrogen atoms and an oxygen atom get close enough, the electron from each atom feels an attraction from the protons in the other atom's nucleus.
- This attraction pulls the atoms together.
- The electrons end up being shared by the atoms in a region around the nucleus of both atoms.
Covalent Bonding in Water II --> oceanography class was in session
--> the COLE Hall oceanography classroom for U.S.Navy intellectuals regarding the U.S.S.Cole of October 2000 and their university THINK TANK errors in the simple explanation.

Hydrogen atoms and an oxygen atom are close together.
Electrons from each atom feel the attraction from the protons in the nucleus of the other atom.
This attraction pulls the atoms together and the electrons are shared by both atoms.
The
atoms bond because there is a strong enough attraction in both
directions and room for the electrons in the outer energy level of the
atoms.
The above water molecule ..... is shown below ... in the WATER geometry using the concrete/cement model of the univeristy campus builfing Cole Hall.
Above, we had the H and H symbols of the Hydogen atom represented in physical FORMAT VIA the larger sized ..... cement/concrete/brick builkfing lecture HALLS ..... lecture HALL 1 and lecture HALL 2 of Cole Hall.
Tto complete the water molecule design configuration ... we need an O = Oxygen .... a cirlce or round shape near Cole Hall.
The picture below shows the O = Oxygen patallel in the round driveway /walkway in front of the COLE HALL building ( the Hydrogen Halls).
The oceanography class provides additional information about the super-symmetry physics parallel of this situation.
Images for covalent bond water molecule
Introduction to Supersymmetry - Hitoshi Murayama
Supersymmetry: Looking in Nature's Mirror « NOVA's Physics Blog ...
Physicists Debate Future of Supersymmetry | Simons Foundation
the cement/brick buildging inside ( H and H) and the outside round driveway (O = Oxygen)
with the additional CLUES of
- the scene at Cole Hall, while Daniel Parmenter was pronounced dead implies .....
Daniel Parmenter --> Da Parm ---> Data Parametere
Parameter | Define Parameter at Dictionary.com
Daniel Parameters - US Army War College --> Northern Illinois University
Data Parameters - United States (States of MIND) VERSUS Colleges and Universities
Parameters - US Army War College
Parameters - US Army War College
Par me ter - US Army War Col
Parmenter - US Army War Cole HALL
Supersymmetry, Grand Unification, and String Theory - iTunes - Apple
Images for covalent bond water molecule
Images for co valent bond water molecule
Images for cole valentine water molecule mass expression
Northern Ill / noise Universe shooting - Wikipedia, the free encyclopedia
GCSE CHEMISTRY - Covalent Bonding in a Water Molecule - What ...
An oxygen atom has 6 electrons in its outer shell. Oxygen is in group 6 ...
Bulletin of the Atomic Scientists:
Images for human water
Covalent Bonding in a Water objects --> Water MAN

We see the important relationship beween different levels ...different perceptions of REALITY ..
Now let's look at the astrophysics VIEW of the UNitred States geography regions and its universities, campus buildings, Nature's SYMBOL MACHINEs that use English language alphabets and periodic atomic table symbols, college textbook information formula, humnan activity, etc .
THe astrophysics VIEW (like a NASA astronaut in a space station) sees EARTH and see the building below ...with the O = Region of Convergence (ROC) as used in signal processing analysis.
THus the SYMBOL MACHINE analysis of cement/concrete building label: Cole Hall and the O of the cement /concrete driveway gives ---> CHO.
Astrophysics chemistry sees symbol CO --> CO.valent social chemsitry bonf at new technology site ...DARPA virgin science at Virginia TECH ...and sees a MR,CHO........ Nature's Darwinianian selection of a covalent bond partner to complete the connection beween the 2 educational institutions.
Virginia Tech massacre - Wikipedia, the free encyclopedia
The Virginia Tech massacre was a school shooting that took place on April 16, 2007, on the campus of Virginia Polytechnic Institute and State University in Blacksburg, Virginia, United States. Seung-Hui Cho, a senior at Virginia Tech, shot and killed 32 people.
two separate attacks, approximately two hours apart --> data that helps describe the covalent bond of the 2 schools at 2 different levels of Sartre existential expression.
Types of Bonds - Properties of Nonmetals - Boundless
while covalent bonds form when electrons are shared between two nonmetals.
Covalent Compounds - Elmhurst College
Using the Wave Theory, the covalent bond
Northern Illinois University, Cole Hall DeKalb Feb 2007 .
Virginia
TECH shooting in April 2007 ........... AP = APRIL, and AP =
Astro.Physics events with the Hawking EVENT HORIZON --->
transformed to ----> EVENT HORROR that rises up and displays a tragic message.
We ought try to understand the SIGNAL about existence and the conflicts among dimensions in our atomic/molecular daily lives.
11th dimension - Wikipedia, the free encyclopedia
Astrophysics VIEW of EARTH and the water molecules ..... continuum: oceans, lakes, people, Navy, ocean class, hydrology,etc.
Given
the outline above, we see that the architecture of existence on EARTH
is very complex and compose of a wide variety of objects and formats
and expressions. Thus EARTH space / time is contains many physical
things: people, buildings , cities, farms, forest,etc.
In
addition, it now contains symbol buildings: languages for the
eye/optical nerve. A college textbook in biochemistry is a symbol
building with special words, data, cell process diagrams,
flowcharts, etc.
Ten thousand years ago the biochemistry molecules existed in physical biology ,,,,, but did not have symbol existence.
Thus
...in the last 100 years ..the last 50 years...since 1960 ... these
molecules experienced Darwinian evolution into the world of
symbols, nouns, flowcharts, etc. Now, in year 2103 we can perceive
...sometimes...that a molecule as 2 identities .... especially since
the biochemistry textbooks are emerging into the James Joyce data
stream of consciousness ... that influences thought and behavior.
Now, the astrophysics VIEW of EARTH is "The Tale of TWO CITIES" ..... physical buildings and symbol.s buildings.
Using general concepts ...we have
1) Cole Hall ..Oceanography ---> capital letter initials ----> CHO extensions into the symbol worlds -->
George Orwell and book 1984 and Oceania and propaganda AND physics wave mechanics and EM propagation waves
The HALL event CHO occurred on Feb 14, 2007
2) The 2nd HALL event CHO occurred on April 16, 007 at Norris engineering HALL with MR.CHO with English language engineering, social engineering, and the Virginia home of the .... Constitution of the UNITED STATES engineering.
Thus we see the convergence of these different engineering systems at Norris Hall at Virginia TECH.
Hydrology - Wikipedia, the free encyclopedia
Virginia Tech massacre - Wikipedia,
About two hours after the initial shootings, CHO entered Norris Hall, which
About two hours after the initial shootings, Cole Hall Oceanography class entered Norris Hall for the advanced hydrology class..
Thus we see a few pieces to a puzzle --->
Career Opportunities - U.S. Naval Research Laboratory